CBSE Class 12 Chemistry Answer Key 2025: The Central Board of Secondary Education (CBSE) conducted the Class 12 Business Studies examination on February 27, 2025. Following the exam, students are eager for their performance. To help them in this process, we provide the answer keys for all sets of the question paper in PDF format.
CBSE Class 12 Chemistry Answer Key Features
Utilising the answer key offers several benefits:
- Self-Assessment: Helps evaluate your performance and understanding of Chemistry concepts.
- Clarification of Doubts: Provide clear solutions to numerical and theoretical questions you found challenging.
- Preparation for Future Exams: Identifies areas where more practice is needed, helping you prepare better for upcoming board and competitive exams like NEET and JEE.
Check| CBSE Class 12 Chemistry Question Paper 2025 (All Sets)
CBSE Class 12 Chemistry Paper Analysis 2025 (All Sets)
CBSE Class 12 Chemistry Exam Pattern and Marking Scheme 2025
| S.No. | Title | Marks |
| 1 | Solutions | 7 |
| 2 | Electrochemistry | 9 |
| 3 | Chemical Kinetics | 7 |
| 4 | d -and f -Block Elements | 7 |
| 5 | Coordination Compounds | 7 |
| 6 | Haloalkanes and Haloarenes | 6 |
| 7 | Alcohols, Phenols and Ethers | 6 |
| 8 | Aldehydes, Ketones and Carboxylic Acids | 8 |
| 9 | Amines | 6 |
| 10 | Biomolecules | 7 |
|
| Total | 70 |
CBSE Class 12 Chemistry Exam Pattern 2025 Key Highlights
| Section | Question Type | Number of Questions | Marks per Question | Total Marks |
| SECTION A | Multiple Choice Questions | 16 | 1 | 16 |
| SECTION B | Short Answer Questions | 5 | 2 | 10 |
| SECTION C | Short Answer Questions | 7 | 3 | 21 |
| SECTION D | Case-Based Questions | 2 | 4 | 8 |
| SECTION E | Long Answer Questions | 3 | 5 | 15 |
CBSE Class 12 Chemistry Exam Answer Key- Highlights
The CBSE Class 12 Chemistry exam typically consists of around 30-35 questions, varying based on the latest pattern.
| Feature | Highlights |
| Board Name | Central Board Of Secondary Education (CBSE) |
| Exam Name | CBSE Class 12th Chemistry Exam |
| Exam Date | February 27, 2025 |
| Duration | 10:30 AM to 1:30 PM (3 hours) |
| Total Marks | 70 |
| Mode | Offline (Pen and Paper) |
| Time Duration | 3 Hours |
| Question Paper Sets: | Multiple Sets (Set 1, Set 2, Set 3) |
CBSE Class 12th Chemistry Answer Key 2025
CBSE Class 12th Chemistry Set-1 Answer Key
Get here the CBSE Class 12th Chemistry Answer Key 2025 which is curated by the subject matter experts. Students can access the answers to all questions asked in the CBSE Class 12th Chemistry Question Paper 2025.
SECTION - A
16 × 1 = 16
Questions No. 1 to 16 are Multiple Choice type questions carrying 1 mark each.
1. The charge required for the reduction of 1 mol of MnO4−to MnO2 is
(A) 1 F
(B) 3 F
(C) 5 F
(D) 6 F
Correct Answer: (B) 3 F
2. Which among the following is a false statement?
(A) Rate of zero-order reaction is independent of the initial concentration of the reactant.
(B) Half-life of a zero-order reaction is inversely proportional to the rate constant.
(C) Molecularity of a reaction may be zero.
(D) For a first-order reaction, t1/2= 0.693/k.
Correct Answer: (C) Molecularity of a reaction may be zero.
3. The number of molecules that react with each other in an elementary reaction is a measure of the
(A) the activation energy of the reaction
(B) stoichiometry of the reaction
(C) the molecularity of the reaction
(D) order of the reaction
Correct Answer: (C) molecularity of the reaction
4. The element having [Ar]3d104s2 electronic configuration is
(A) Cu
(B) Zn
(C) Cr
(D) Mn
Correct Answer: (B) Zn
5. The complex ions [Co(NH3)5(NO2)]2+and [Co(NH3)5(ONO)]2+ are called
(A) Ionization isomers
(B) Linkage isomers
(C) Co-ordination isomers
(D) Geometrical isomers
Correct Answer: (B) Linkage isomers
6. The diamagnetic species is:
(A) [Ni(CN)4]2-
(B) [NiCl4]2−
(C) [Fe(CN)6]3−
(D) [CoF6]3−
Correct Answer: (A) [Ni(CN)4]2-
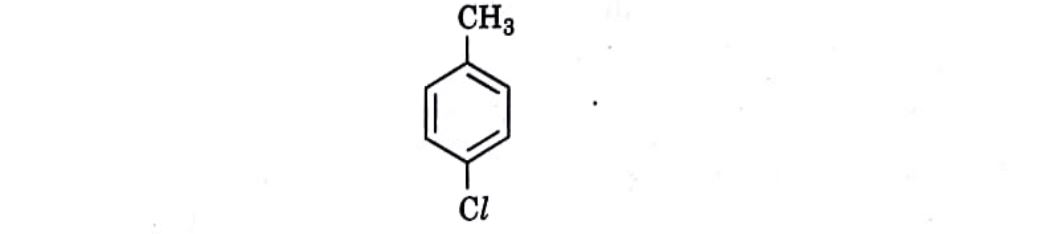
7. Which is the correct IUPAC name for the given compound?
(A) Methylchlorobenzene
(B) Toluene
(C) 1-Chloro-4-Methylbenzene
(D) 1-Methyl-4-Chlorobenzene
Correct Answer: (C) 1-Chloro-4-Methylbenzene
8. What will be formed after the oxidation reaction of secondary alcohol with chromic anhydride (CrO3)?
(A) Aldehyde
(B) Ketone
(C) Carboxylic acid
(D) Ester
Correct Answer: (B) Ketone
9. The conversion of phenol to salicylic acid can be accomplished by
(A) Reimer-Tiemann reaction
(B) Friedel-Crafts reaction
(C) Kolbe reaction
(D) Coupling reaction
Correct Answer: (A) Reimer-Tiemann reaction
10. Which of the following is/are examples of denaturation of protein?
(A) Coagulation of egg white
(B) Curdling of milk
(C) Clotting of blood
(D) Both (A) and (B)
Correct Answer: (D) Both (A) and (B)
11. Nucleotides are joined together by
(A) Glycosidic linkage
(B) Peptide linkage
(C) Hydrogen bonding
(D) Phosphodiester linkage
Correct Answer: (D) Phosphodiester linkage
12. Scurvy is caused due a deficiency of
(A) Vitamin B1
(B) Vitamin B2
(C) Ascorbic acid
(D) Glutamic acid
Correct Answer: (C) Ascorbic acid
For questions numbers 13 to 16, two statements are given – one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (A), (B), (C), and (D) as given below:
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
13. Assertion (A): In a first-order reaction, if the concentration of the reactant is doubled, its half-life is also doubled.
Reason (R): The half-life of a reaction does not depend upon the initial concentration of the reactant in a first-order reaction.
Correct Answer: (D) Assertion (A) is false, but Reason (R) is true.
14. Assertion (A): Cu cannot liberate H2 in reaction with dilute mineral acids.
Reason (R): Cu has a positive electrode potential.
Correct Answer: (A) Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of the Assertion (A).
15. Assertion (A): Aromatic primary amines cannot be prepared by Gabriel Phthalimide synthesis.
Reason (R): Aryl halides do not undergo nucleophilic substitution reaction with the anion formed by phthalimide.
Correct Answer: (A) Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of the Assertion (A).
16. Assertion (A): Vitamin D cannot be stored in our body.
Reason (R): Vitamin D is a fat-soluble vitamin and is not excreted from the body in urine.
Correct Answer: (D) Assertion (A) is false, but Reason (R) is true.
CBSE Class 12th Chemistry Set-3 Answer Key
1. Which among the following is a false statement?
(A) Rate of zero-order reaction is independent of the initial concentration of the reactant.
(B) Half-life of a zero-order reaction is inversely proportional to the rate constant.
(C) Molecularity of a reaction may be zero.
(D) For a first-order reaction, t1/2= 0.693/k.
Correct Answer: (C) Molecularity of a reaction may be zero.
2. The charge required for the reduction of 1 mol of MnO4−to MnO2 is
(A) 1 F
(B) 3 F
(C) 5 F
(D) 6 F
Correct Answer: (B) 3 F
3. The element having [Ar]3d104s2 electronic configuration is
(A) Cu
(B) Zn
(C) Cr
(D) Mn
Correct Answer: (B) Zn
4. The number of molecules that react with each other in an elementary reaction is a measure of the :
(A) Activation energy of the reaction
(B) Stoichiometry of the reaction
(C) Molecularity of the reaction
(D) Order of the reaction
Correct Answer: (C) Molecularity of the reaction
5. The diamagnetic species is:
(A) [Ni(CN)4]2-
(B) [NiCl4]2−
(C) [Fe(CN)6]3−
(D) [CoF6]3−
Correct Answer: (A) [Ni(CN)4]2-
6. The complex ions [Co(NH3)5(NO2)]2+and [Co(NH3)5(ONO)]2+ are called
(A) Ionization isomers
(B) Linkage isomers
(C) Co-ordination isomers
(D) Geometrical isomers
Correct Answer: (B) Linkage isomers
7. What will be formed after the oxidation reaction of secondary alcohol with chromic anhydride (CrO3)?
(A) Aldehyde
(B) Ketone
(C) Carboxylic acid
(D) Ester
Correct Answer: (B) Ketone
8. Which is the correct IUPAC name for the given compound?
(A) Methylchlorobenzene
(B) Toluene
(C) 1-Chloro-4-Methylbenzene
(D) 1-Methyl-4-Chlorobenzene
Correct Answer: (C) 1-Chloro-4-Methylbenzene
9. Which of the following is/are examples of denaturation of protein?
(A) Coagulation of egg white
(B) Curdling of milk
(C) Clotting of blood
(D) Both (A) and (B)
Correct Answer: (D) Both (A) and (B)
10. The conversion of phenol to salicylic acid can be accomplished by
(A) Reimer-Tiemann reaction
(B) Friedel-Crafts reaction
(C) Kolbe reaction
(D) Coupling reaction
Correct Answer: (A) Reimer-Tiemann reaction
11. Scurvy is caused due to deficiency of
(A) Vitamin B1
(B) Vitamin B2
(C) Ascorbic acid
(D) Glutamic acid
Correct Answer: (C) Ascorbic acid
12. Nucleotides are joined together by
(A) Glycosidic linkage
(B) Peptide linkage
(C) Hydrogen bonding
(D) Phosphodiester linkage
Correct Answer: (D) Phosphodiester linkage
For questions numbers 13 to 16, two statements are given – one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (A), (B), (C), and (D) as given below:
(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).
(B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of Assertion (A).
(C) Assertion (A) is true, but Reason (R) is false.
(D) Assertion (A) is false, but Reason (R) is true.
13. Assertion (A): In a first-order reaction, if the concentration of the reactant is doubled, its half-life is also doubled.
Reason (R): The half-life of a reaction does not depend upon the initial concentration of the reactant in a first-order reaction.
Correct Answer: (D) Assertion (A) is false, but Reason (R) is true.
14. Assertion (A): Cu cannot liberate H2 in reaction with dilute mineral acids.
Reason (R): Cu has a positive electrode potential.
Correct Answer: (A) Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of the Assertion (A).
15. Assertion (A): Aromatic primary amines cannot be prepared by Gabriel Phthalimide synthesis.
Reason (R): Aryl halides do not undergo nucleophilic substitution reaction with the anion formed by phthalimide.
Correct Answer: (A) Both Assertion (A) and Reason (R) are true, and Reason (R) is the correct explanation of the Assertion (A).
16. Assertion (A): Vitamin D cannot be stored in our body.
Reason (R): Vitamin D is a fat-soluble vitamin and is not excreted from the body in urine.
Correct Answer: (D) Assertion (A) is false, but Reason (R) is true.
We are adding some more answers. Keep refreshing the article.
CBSE Class 12th Chemistry Answer Key 2025 (Link To Be Active Soon) |
Other Related Links
Comments
All Comments (0)
Join the conversation