CBSE Class 10 Science paper in term 2 exam will have case study questions of 8 marks in Section C. To help students score full marks in this section, we have provided below important case study questions that has been prepared by the subject experts who carry a tremendous experience in the teaching field. These questions have been prepared specially for the CBSE Term 2 Exam, considering the latest pattern and reduced syllabus. Students must practice the chapter-wise important questions provided below for a quick last minute revision and increase their chances of scoring high in the CBSE Class 10 Science Term 2 Exam 2022.
| CBSE Class 10 Science Important 2 Marks Questions for Term 2 Exam |
| CBSE Class 10 Science Important 3 Marks Questions for Term 2 Exam |
Check some of the important case study questions below:
Q. Read the following carefully and answer the questions.
In covalent compounds atoms share valence electrons to satisfy the octet. Each atom shares one pair or two pairs or three pairs of electrons depending on their combining capacity. In electron dot structures only number of valence electrons are shown around the symbols of constituent atoms. Carbon using its valency of four can make either single, double or triple bonds with other carbon atoms or any other atoms. Carbons self-linking property is called catenation. In hydrocarbons carbon makes aliphatic or cyclic molecules they are either saturated or unsaturated. Based on these facts Read the following paragraph and answer the questions given below.
An element X combines with Y to form a colourless odourless gas, Z which turns lime water milky is the major constituent of all organic molecules. Five X atoms combines with hydrogens to form a cyclic saturated hydrocarbon J and aliphatic unsaturated hydrocarbon Q. Q is used in gas welding.
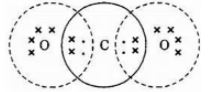
(1) Identify compound Z and draw its electron dot structure.
(2) Write the chemical formula and IUPAC name of compound Q
(3) What is the common name of Q
(4) How many single covalent bonds are present in compound J?
Answer:
1) Z is CO2 its electron dot structure is
2) C2H2, ethyne
3) Acetylene
4) 15
Q. Read the following carefully and answer the questions.
Atoms of eight elements A, B, C, D, E, F, G and H have the same number of shells but different number of electrons in their outermost shell. It was found that elements A and G combine to form an ionic compound. This compound is added in a small amount to almost all vegetable dishes during cooking. Oxides of elements A and B are basic in nature while those of E and F are acidic. The oxide of D is almost neutral.
Based on the above information answer the following questions:
(1) To which group or period of the Periodic Table do the listed elements belong?
(2) What would be the nature of compound formed by a combination of elements B and F?
(3) Which two of these elements could definitely be metals?
(4) If the number of electrons in the outermost shell of elements C and G be 3 and 7 respectively, write the formula of the compound formed by the combination of C and G.
Answer:
1. A and B belong to group 1 and 2 because they form basic oxides.
C belongs to group 13 as it has 3 valence electrons.
D belongs to group 14 as it forms almost neutral oxide.
E and F belong to group 15 and 16 as they form acidic oxides,
G belongs to group 17 as it has 7 valence electrons and
H belongs to group 18.
They belong to 3rd period of the Periodic Table because AG is NaCl, added in a small amount to almost all vegetable dishes during cooking and Na and Cl belong to 3rd period.
2. Ionic compounds will be formed because ‘B’ is metal and ‘F’ is non-metal. ‘B’ can lose two electrons and ‘F’ can gain two electrons.
3. A and B are definitely metals as they form basic oxides.
4. CG3 is the formula of the compound formed by combination of C and G.
Download the chapter-wise case-based questions from the following link:
| CBSE Class 10 Science Important Case Study Questions for Term 2 Exam 2022 |
Also, check CBSE Class 10 Science Best Study Material for Term 2 Exam 2022
Comments
All Comments (0)
Join the conversation