CBSE Board Exam 2022 (Term 2): Get here the CBSE term 2 sample paper and marking scheme for Class 10 Science. The CBSE Class 10 Science Term 2 Sample Paper 2022 reveals the design of the question paper for the Science Term 2 Exam that will be held on May 10, 2022. CBSE Class 10 Science Marking Scheme 2022 depicts the answer hints that can be considered as near to solutions to all questions given in the sample paper. The CBSE Marking Scheme also helps to understand how to write concise and step-wise solutions in the board exam.
Check below CBSE Class 10 Science (086) Sample Paper Term 2 (2022):
Max. Marks: 40
Time allowed: 2 hours
General Instructions:
i) All questions are compulsory.
ii) The question paper has three sections and 15 questions. All questions are compulsory.
iii) Section–A has 7 questions of 2 marks each; Section–B has 6 questions of 3 marks each; and Section–C has 2 case based questions of 4 marks each.
iv) Internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
Also, Check CBSE Class 10 Science Term 2 Syllabus 2022
Section - A
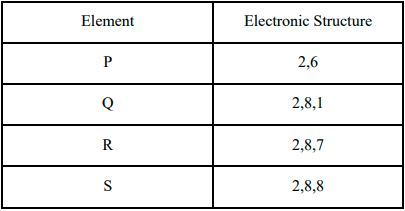
1. The table shows the electronic structures of four elements.
a. Identify which element(s) will form covalent bonds with carbon.
b. “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
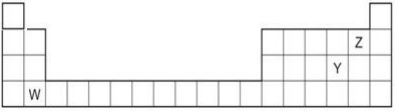
2. The diagram below shows part of the periodic table.
a. Which elements would react together to form covalent compounds?
b. Between the two elements W and Z, which will have a bigger atomic radius? Why?
3. a. Trace the path a male gamete takes to fertilise a female gamete after being released from the penis.
b. State the number of sets of chromosomes present in a zygote.
4. Rajesh observed a patch of greenish black powdery mass on a stale piece of bread.
a. Name the organism responsible for this and its specific mode of asexual reproduction.
b. Name its vegetative and reproductive parts.
5. Mustard was growing in two fields- A and B. While Field A produced brown coloured seeds, field B produced yellow coloured seeds.
It was observed that in field A, the offsprings showed only the parental trait for consecutive generations, whereas in field B, majority of the offsprings showed a variation in the progeny. What are the probable reasons for these?
OR
In an asexually reproducing species, if a trait X exists in 5% of a population and trait Y exists in 70% of the same population, which of the two trait is likely to have arisen earlier? Give reason.
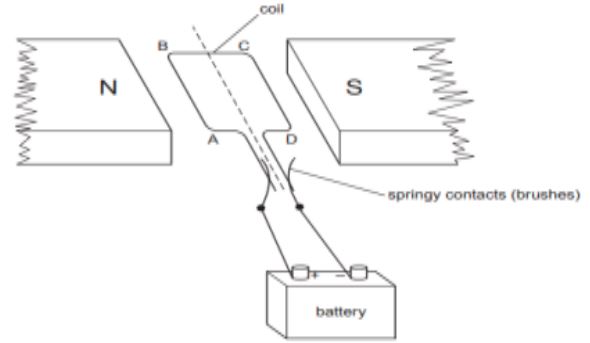
6. A simple motor is made in a school laboratory. A coil of wire is mounted on an axle between the poles of a horseshoe magnet, as illustrated.
In the example above, coil ABCD is horizontal and the battery is connected as shown.
a. For this position, state the direction of the force on the arm AB.
b. Why does the current in the arm BC not contribute to the turning force on the coil?
OR
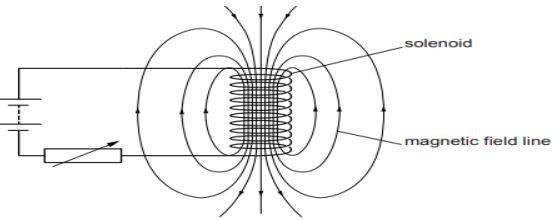
A circuit contains a battery, a variable resistor and a solenoid. The figure below shows the magnetic field pattern produced by the current in the solenoid.
a. State how the magnetic field pattern indicates regions where the magnetic field is stronger.
b. What happens to the magnetic field when the current in the circuit is reversed?
7. DDT was sprayed in a lake to regulate breeding of mosquitoes. How would it affect the trophic levels in the following food chain associated with a lake? Justify your answer.
OR
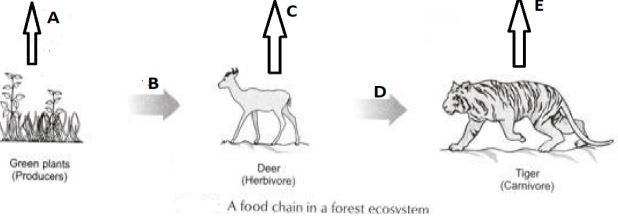
In the following food chain, vertical arrows indicate the energy lost to the environment and horizontal arrows indicate energy transferred to the next trophic level. Which one of the three vertical arrows (A, C and E) and which one of the two horizontal arrows (B and D) will represent more energy transfer? Give reason for your answer.
Section - B
8. Choose an element from period 3 of modern periodic table that matches the description given below in each instance. Give reason for your choice.
a. It has a similar structure to diamond.
b. It has same valency as Lithium. c. It has variable valency and is a member of the Oxygen family (group 16).
9. a. How many isomers are possible for the compound with the molecular formula C4H8? Draw the electron dot structure of branched chain isomer.
b. How will you prove that C4H8 and C5H10 are homologues?
OR
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
a. Identify ‘A’ and draw its electron dot structure.
b. Give the molecular formulae of any two homologues of ‘A’.
10. Two pea plants - one with round yellow seeds (RRYY) and another with wrinkled green (rryy) seeds produce F1 progeny that have round, yellow (RrYy) seeds.
When F1 plants are self-pollinated, which new combination of characters is expected in F2 progeny? How many seeds with these new combinations of characters will be produced when a total 160 seeds are produced in F2 generation? Explain with reason.
Download the complete CBSE Class 10 Term 2 Sample Paper and its CBSE Marking Scheme from the following links:
| CBSE Class 10 Science Sample Paper for Term 2 Exam 2022 (PDF) |
| CBSE Class 10 Science Marking Scheme of Sample Paper 2022 (PDF) |
Also Check:
CBSE Class 10 Science Best Study Material for Term 2 Exam 2022

Comments
All Comments (0)
Join the conversation