CBSE Practice Paper 2021-22: CBSE Class 10 Science Term 2 Paper will be conducted on 10th May, 2022. Right now is the time to revise all that you have studied during your exam preparations. To do this effectively, solve as many practice papers and sample papers as possible. Considering the need of the hour, Jagran Josh presents here an exclusive practice paper for CBSE Class 10th to help students gear up their preparations for the Term 2 Exam. This practice paper is set by experienced teachers. This is entirely based on the pattern of the latest CBSE Class 10 Science Term 2 Sample Paper. The solution of the practice paper is also made available for the convenience of students. Both the practice papers and solutions can be downloaded in PDF format.
New! CBSE Class 10 Science Term 2 Paper Writing & Time Management Tips by Experts
CBSE Class 10 Science Practice Paper for Term 2 Exam 2022:
Max. Marks: 40
Time allowed: 2 hours
General Instructions:
i) All questions are compulsory.
ii) The question paper has three sections and 15 questions. All questions are
iii) Section–A has 7 questions of 2 marks each; Section–B has 6 questions of 3 marks each; and Section–C has 2 case-based questions of 4 marks each.
iv) Internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
Also Check
CBSE Class 10 Science Best Last Minute Revision Tips By Experts for Term 2 Exam 2022
CBSE Class 10 Science Paper Pattern and Chapter-wise Weightage for Term 2 Exam 2022
Section A
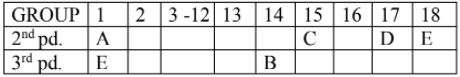
1. Study the table given below carefully and answer the questions.
(a) Which element is a nonmetal with valency 1?
(b) Which element makes only covalent bond?
(c) Which element has the largest atomic radius?
(d) Write the electronic configuration of the element C
2. How does binary fission differ from multiple fission? Give one difference between the binary fission in amoeba and leishmania?
3. Woman had undergone surgical method for preventing unwanted pregnancies. Does this protect her from getting sexually transmitted diseases? Write any other two contraceptive methods.
4.Write any two limitations of Mendeleev’s periodic table and how these defects are rectified in Modern periodic table?
5. Explain the mechanism by which the chromosome number is maintained constant in human beings?
OR
With the help of suitable example, explain characters may be inherited, but may not be expressed always.
6. A coil of insulated copper wire is connected to a galvanometer. What will happen if a bar magnet is
(a) pushed into the coil?
(b) withdrawn from inside the coil?
Give reason also.
OR
Name any 2 factors on which the strength of the magnetic field produced by a solenoid depends? How does it depend on these factors?
7. Explain with the help of an example, why the number of trophic levels in a food chain is limited?
OR
A food chain consists of fish, larvae, phytoplankton and birds. The level of pesticides in water in which these organisms live is high.
(a) Which organism will have the maximum amount of pesticides accumulated through the food chain and what is this process known as?
(b) Write the food chain comprising all the organisms mentioned above.
Download the complete paper and its solution from their respectice links:
CBSE Class 10 Science Practice Paper for Term 2 Exam 2022 | Solution of Practice Paper |
Comments
All Comments (0)
Join the conversation