CBSE Class 12 Chemistry Sample Question Paper 2022-23: CBSE board has released the Chemistry Sample Paper and marking scheme for 2022-23 bringing the candidates closer to their board exams.
Although there’s a whole lot of hype about how difficult boards can get, there’s nothing to worry about. If studied consistently from the right resources, you will surely perform your best.
First of all, you should ensure to have the whole syllabus in order to not miss any chapter or topic. You will find the Chemistry syllabus for CBSE class 12th here.
If you are looking for the right resources to study from, click here to get the complete chapter-wise chemistry notes based on prescribed NCERT course content.
Moving on to the latest sample paper by the board.
The sample paper starts with the general instructions.
The general instructions provided in the CBSE Class 12 Chemistry Sample Question Paper 2022-23 for the students to follow are:
Maximum Marks :70
Time allotted: 3 hours
General Instructions:
Read the following instructions carefully.
- a) There are 35 questions in this question paper with internal choice.
- b) SECTION A consists of 18 multiple-choice questions carrying 1 mark each.
- c) SECTION B consists of 7 very short answer questions carrying 2 marks each.
- d) SECTION C consists of 5 short answer questions carrying 3 marks each.
- e) SECTION D consists of 2 case- based questions carrying 4 marks each.
- f) SECTION E consists of 3 long answer questions carrying 5 marks each.
- g) All questions are compulsory.
- h) Use of log tables and calculators is not allowed
Candidates can check the CBSE Class 12 Chemistry Sample Question Paper 2022-23 below to prepare well for their CBSE Class 12 Board Exam 2022-23.
SECTION A
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
1. The major product of acid catalysed dehydration of 1-methylcyclohexanol is:
a. 1-methylcyclohexane
b. 1-methylcyclohexene
c. 1-cyclohexylmethanol
d. 1-methylenecyclohexane
2. Which one of the following compounds is more reactive towards SN1 reaction?
a. CH2=CHCH2Br
b. C6H5CH2Br
c. C6H5CH (C6H5)Br
d. C6H5CH(CH3) Br
3. KMnO4 is coloured due to:
a. d-d transitions
b. charge transfer from ligand to metal
c. unpaired electrons in d orbital of Mn
d. charge transfer from metal to ligand
4. Which radioactive isotope would have the longer half- life 15O or 19O? (Given rate constants for 15O and 19O are 5.63x 10-3 s-1 and k = 2.38 x 10-2s-1 respectively.)
a. 15O
b. 19O
c. Both will have the same half-life
d. None of the above, information given is insufficient
5. The molar conductivity of CH3COOH at infinite dilution is 390 Scm2/mol. Using the graph and given information, the molar conductivity of CH3COOK will be
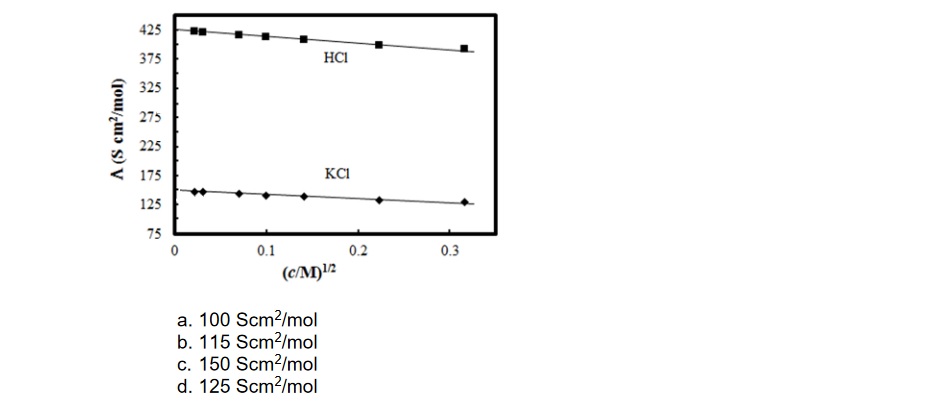
We have shared the CBSE Class 12 Chemistry Sample Paper 2022-23 in pdf format below, if you want to download and print it for better access to the hard copy.
Download the CBSE Class 12 Chemistry Sample Paper 2022-23 in pdf format
The marking scheme of the CBSE Class 12 Chemistry Sample Paper 2022-23 in pdf format is attached below for you to study accordingly.
Download the marking scheme of CBSE Class 12 Chemistry Sample Paper 2022-23 in pdf format
Using the CBSE Class 12 Chemistry Sample Paper 2022-23 and with the help of other resources such as NCERT textbook pdf, NCERT solutions, Previous year papers and marking scheme, students can practise for the Chemistry theory board exams.
Also, you should stay careful about the Chemistry practical exams. Students often face dull results because they did not prepare well for C chemistry theory papers. This is because students get lost in the vast syllabus of the theory portions. To cover the syllabus of CBSE class 12 Chemistry practicals, you should click here.
Stay tuned to Jagran Josh for more content related to CBSE Class 12 board exams 2022-23.
Evaluate yourself using the link below:
Best of luck to all the candidates.
Comments
All Comments (0)
Join the conversation