Check CBSE Science answer key 2021-22 of Term 1 CBSE Class 10 board exam 2021-22 (Term 1) along with question paper & reviews. CBSE Answer Key 2021-22 for Class 10 Science board exam 2021-22 (Term 1) question paper has been verified by CBSE School teachers. The PDF of the CBSE Class 10th Science question paper (Term 1 CBSE board exam 2021-22) has been provided by Jagran Josh along with the review & feedback of students. One can access paper and reviews from the links given below.
CBSE Answer Key: 10th Maths Term 1 (Advanced/Standard) CBSE Board Exam 2021-22
CBSE Answer Key: 10th Maths (Basic) Term 1 CBSE Board Exam 2021-22
CBSE Class 10 Maths Term 1 Exam Analysis: Moderately tough paper with a few tricky questions; Students feedback, Answer Key here
CBSE Class 10 Science Term 1 Exam Analysis (by Experts) & Students’ Feedback
Also Check: CBSE Class 10 Maths Important Questions for Board Exam 2021 (with Solutions)
CBSE Class 10 Science Answer Key 2021-22 (Term 1): CBSE Board Exam 2021-22
1. A student took Sodium Sulphate solution in a test tube and added Barium Chloride solution to it. He observed that an insoluble substance has formed. The colour and molecular formula of the insoluble substance is
(a) Grey, Ba2SO4
(b) Yellow, Ba(SO4)2,
(c) White, BaSO4
(d) Pink, BaSO4,
Answer: (c) White, BaSO4
2. Which of the following oxide(s) is/are soluble in water to form alkalis?
(i) Na2O (ii) SO2 (iii) K2O (iv) NO2
(a) (i) and (iii)
(b) (i) only
(c) (ii) and (iv)
(d) (iii) only
Answer: (a)
3. Study the diagram given below and identify the gas formed in the reaction.
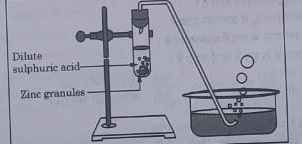
(a) Carbon di-oxide which extinguishes the burning candle.
(b) Oxygen due to which the candle burns more brightly.
(c) Sulphur dioxide which produces a suffocating smell.
(d) Hydrogen which while burning produces a popping sound.
Answer: (d)
4. Sodium reacts with water to form sodium hydroxide and hydrogen gas. The balanced equation which represents the above reaction is
(a) Na(s) + 2H20(l) → 2NaOH(aq) + 2H2(g)
(b) 2Na(s) + 2H2O(l) → 2NaOH(aq) +H2(g)
(c) 2Na(s) + 2H20(l) → NaOH(aq) + 2H2(g)
(d) 2Na(s) + H20(l) - 2NaOH(aq) + 2H2(g)
Answer: (b)
5. Which of the options in the given table are correct?
Option | Natural Source | Acid Present
(i) Orange | Oxalic acid
(ii) Sour milk | Lactic acid
(iii) Ant sting | Methanoic acid
(iv) Tamarind | Acetic acid
(a) (i) and (ii)
(b) (i) and (iv)
(c) (ii) and (iii)
(d) (ii) and (iv)
Answer: (c)
6. C6H12O6 (aq) + 602(aq) →6CO2(aq) + 6H2O
The above reaction is a/an
(a) displacement reaction
(b) endothermic reaction
(c) exothermic reaction
(d) neutralisation reaction
Answer: (c)
7. Which of the following statements about the reaction given below are correct?
MnO2 + 4HCI → MnCl2 +2H20 + Cl2
(i) HCl is oxidized to Cl2
(ii) MnO2 is reduced to MnCl2
(iii) MnCl2 acts as an oxidizing agent
(iv) HCl acts as an oxidizing agent
(a) (i), (iii) and (iv)
(b) (i), (ii) and (iii)
(c) (i) and (ii) only
(d) (iii) and (iv) only
Answer: 7. (c)
8. Select from the following the statement which is true for bases.
(a) Bases are bitter and turn blue litmus red.
(b) Bases have a pH less than 7
(c) Bases are sour and change from red litmus to blue.
(d) Bases turn pink when a drop of phenolphthalein is added to them.
Answer: (d) Bases turn pink when a drop of phenolphthalein is added to them.
9. Study the following table and choose the correct option:
Salt - Parent Acid - Parent Base - Nature of Salt
(a) Soidum Chloride - HCI - NaOH - Basic
(b) Sodium Carbonate - H2CO3 - NaOH - Neutral
(c) Sodium Sulphate - H2SO4 - NaOH - Acidic
(d) Sodium Acetate - CH3COOH - NaOH - Basic
Answer: (d) Sodium Acetate - CH3COOH - NaOH - Basic
10. It is important to balance the chemical equations to satisfy the law of conservation of mass. Which of the following statements of the law is incorrect?
(a) The total mass of the elements present in the reactants is equal to the total mass of the elements present in the products.
(b) The number of atoms of each element remains the same, before and
after a chemical reaction.
(c) The chemical composition of the reactants is the same before and after the reaction
(d) Mass can neither be created nor can it be destroyed in a chemical
Reaction.
Answer 10: (c) The chemical composition of the reactants is the same before and after the reaction
11. Consider the following statements in connection with the functions of the blood vessels marked A and B in the diagram of a human heart as shown.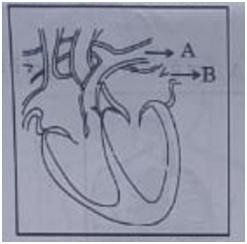
(i) Blood vessel A - It carries Carbon dioxide rich blood to the lungs.
(ii) Blood vessel B - It carries Oxygen rich blood from the lungs.
(iii) Blood Vessel B - Left atrium relaxes as it receives blood from this blood vessel
(iv) Blood Vessel A - Right atrium has thick muscular wall as it has to pump blood to this blood vessel.
The Correct Statements are
(a) (i) and (ii) only
(b) (ii) and (iii) only
(c) (ii), (iii) and (iv)
(d) (i), (ii) and (iii)
Answer: (a)
12. In Living organisms During respiration which of the following Products are not formed if oxygen is not available?
(a) Carbon dioxide + Water
(b) Carbon dioxide + Alcohol
(c) Lactic acid + Alcohol
(d) Carbon dioxide + Lactic Acid
Answer: (b) Carbon dioxide + Alcohol
13. The Correct Statements with reference to single called organisms are
(i) Complex substances are not broken down in to simpler substances.
(ii) Simple diffusion is sufficient to meet the requirement of exchange of gases.
(iii) Specialized tissues perform different functions in the organism.
(iv) Entire surface of the organism is in contact with the environment for taking in food.
(a) (i) and (iii)
(b) (ii) and (iii)
(c) (ii) and (iv)
(d) (i) and (iv)
Answer: (c)
14. Which one among the following is not removed as a waste product from the body of a plant?
(a) Resins and Gums
(b) Urea
(c) Dry Leaves
(d) Excess Water
Answer: (b)
15. Which one of the following statements are correct in reference to the role of A (Shown in the given diagram) during a breathing cycle in human beings?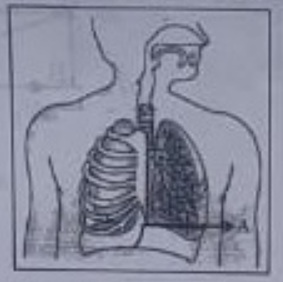
(i) It helps to decrease the residual volume of air in lungs.
(ii) it flattens as we inhale.
(iii) it gets raised as we inhale.
(iv) it helps the chest cavity to become larger.
(a) (ii) and (iv)
(b) (iii) and (iv)
(c) (i) and (ii)
(d) (i), (ii) and (iv)
Answer 15: (d)
16. Which one of the following conditions is true for the state of stomata of a green leaf shown in the given diagram?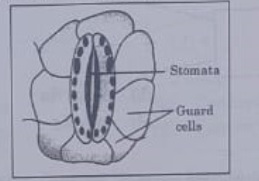
(a) Large amount of water flows in to the ground cells.
(b) Gaseous exchange is occurring in large amount.
(c) Large amount of water flows out from the guard cells.
(d) Large amount of sugar collects in the guard cell
Answer 16: (c)
17. In which of the following is a concave mirror used?
(a) A solar cooker
(b) A rear view mirror in vehicles
(c) A safety mirror in shopping malls
(d) In Viewing full size image of distant tall buildings.
Answer 17: (a)
18. A student wants to obtain magnified image of an object AB as on a Screen. Which one of the following arrangements shows the correct position of AB for him/her to be successful?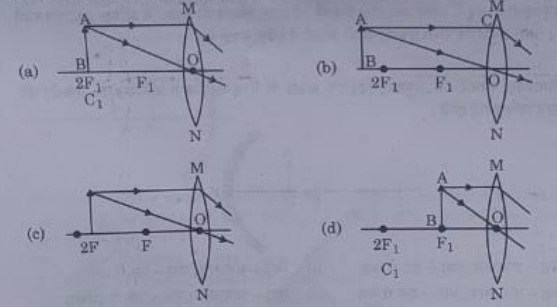
Answer 18: (c)
19. The following diagram shows the use or an optical device to perform an experiment of light. As per the arrangement shown, the optical device is likely to be a;
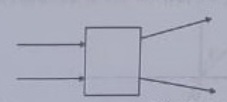
(a) Concave mirror
(c) Convex mirror
(b) Concave lens
(d) Convex lens
Answer 19: (b)
20. A ray of light starting from air passes through medium A of refractive index 1.50, enters medium B of refractive index 1.33 and finally enters medium C of refractive index 2.42. If this ray emerges out in air from C, then for which of the following pairs of media the bending of light least?
(a) air-A
(b) A-B
(c) B-C
(d) C-air
Answer 20: (b)
21. which of the following statements is not true for scattering of light?
(a) Colour of the scattered tight depends on the size of particles of the atmosphere.
(b) Red light is least scattered in the atmosphere.
(c) Scattering of light Lakes place as various colours of white light travel with different speed in fir.
d) The fine particles in the atmospheric air scatter the blue, light more strongly than red. So the scattered blue light enters our eyes.
Answer 21: (c)
22. https://img.jagranjosh.com/images/2021/December/2122021/CBSE-Science-Paper-Term-1-2021-Question-22.jpg
For the diagram shown according to the new Cartesian sign convention the magnification of the image formed will have the following specifications:
(a) Sign - Positive, Value — Leas than 1
(b) Sign — Positive, value - More than 1
(c) Sign — Negative, Value — Less than 1
(d) Sign Negative, value — More than 1
Answer 22: (b)
23.
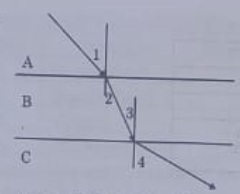
A ray of light is incident as shown. If A, a and C are three different transparent media. then which among the following options is true for the given Diagram?
Answer 23: (c)
24. In the diagram given below X and Y are the end colours of the specturm of white light. The colour of 'Y' Represents the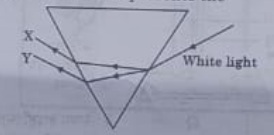
(a) Colour of sky as seen from earth during the day.
(b) colour of the sky as seen from the moon.
(c) Colour used to paint the danger signals.
(d) Colour of sun at the time of noon
Answer 24: (c)
Section - B
Section-B consists of 24 questions (Q. No. 25 to 48). Attempt any 20 questions from this Section. The first attempted 20 questions would be evaluated.
25. which one of the following reactions is categorised as thermal decomposition reaction?
Answer 25: (d)
26. Consider the pH value of the following acidic samples: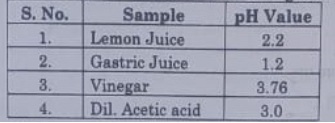
The decreasing order of their H+ ion Concentration is
(a) 3 > 4 > 1 > 2
(b) 2 > 1 > 3 > 4
(c) 2 > 1 > 4 > 3
(d) 3 > 4 > 2 > 1
Answer 26: (c)
27. Study the eperimental set up shown in given figure and choose the correct option from the following:
Answer 27. (d)
28. Which one of the following structures depicts the compound Cacl2?
Answer 28. (d)
29. The pair(s) which will show displacement reaction is/are
(i) NaCl solution and copper metal
(ii) AgNO3 solution and copper metal
(iii) Al2(SO4)3 solution and magnesium metal
(iv) ZnSO4 solution and iron metal
(a) (ii) only
(b) (ii) and (iii)
(c) (iii) and (iv)
(d) (i) and (ii)
Answer 29. (c)
30. Which of the following salts do not have the water of crystalisation?
(i) Bleaching Powder
(ii) Plaster of Paris
(iii) washing soda
(iv) Baking soda
(a) (ii) and (iv)
(b) (i) and (iii)
(c) (ii) and (iii)
(d) (i) and (iv)
Answer 30. (d)
Question No. 31-35 consists of two statements -Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
(a) Both (A) and (R) are true and (R) is the correct explanation of (A).
(b) Both (A) and (R) are true but (R) is not the correct explanation of (A).
(c) (A) is true, but (R) is false.
(d) (A) is false, but (R) is true.
31. Assertion (A) : Sodium hydrogen carbonate is used as an ingredient in antacids.
Reason (R) : NaHCO3 is a mild non-corrosive basic salt.
Answer 31: (b)
32. Assertion (A): Burning of Natural gas is an endothermic process.
Reason (R) : Methane ga combines with oxygen to produce carbon dioxide and water.
Answer 32: (d)
33. Assertion (A) : Nitrogen is an essential element for plant growth and is taken up by plants in the form of inorganic nitrates or nitrites.
Reason (R) : The soil is the nearest and richest source of raw materials like Nitrogen, Phosphorus and other minerals for the plants.
Answer 33: (b)
34. Assertion (A) : Sun appears reddish at the time of Sunrise and Sunset.
Reason (R) : Distance travelled by sunlight in the atmosphere is lesser during sunrise and sunset as compared to noon.
Answer 34: (c)
35. Assertion (A): Hydrochloric acid helps in the digestion of food in the stomach.
Reason (R) : Hydrochloric acid creates an acidic medium to activate protein digesting enzymes.
Answer 35: (a)
36. A student was asked to write a stepwise procedure to demonstrate that carbon dioxide is necessary for photosynthesis. He wrote the following steps. The wrongly worded step is —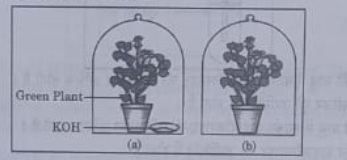
(a) Both potted plants are kept in dark room for at least three days.
(b Bottom of the bell jars is sealed to make them air tight.
(c) Both potted plants are kept in sunlight after the starch test.
(d) A leaf from both the plants is taken to test the presence of starch.
Answer 36: (c)
37. Respiratory structures of two different animals-a fish and a human being are as shown.
observe (a) and (b) and select one characteristic that holds true for both of them.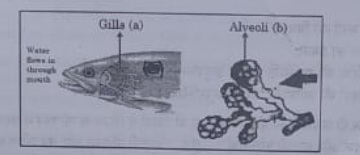
(a) Both are placed internally in the body animal.
(b) Both have thin and moist surface for gaseous exchange.
(c) Both are poorly supplied with blood vessels to conserve energy.
(d) In both the blood returns to the heart after being oxygenated.
Answer 37: (b)
38. observe the diagram of an activity given below. what does it help to conclude, when the person exhales into the test-tube?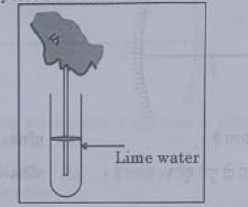
(a) Percentage of carbon dioxide is more in inhaled air.
(b) Fermentation occurs in the presence of oxygen.
(c) Percentage of carbon dioxide is more in the exhaled air.
(d) Fermentation occurs in the presence of carbon dioxide.
Answer 38: (c)
39. If a lens can converge the sun rays at a point 20 cm. away from its optical centre, the power of this lens is —
(a) + 2D
(b) - 2D
(c) + 5D
(d) - 51D
Answer 39: (c)
40. The radius of curvature of a converging mirror is 30 cm. At what distance from the mirror should an object be placed so as to obtain a virtual image?
(a) infinity
(b) 30 cm
(c) Between 16 cm and 30 cm
(d) Between 0 cm and 15 cm
Answer 40: (d)
41. The length of small intestine in a deer is more as compared to the length of small intestine of a tiger. The reason for this is —
(a) Mode of intake of food.
(b) Type of food consumed.
(c) Presence or absence of villi in intestines.
(d) Presence or absence of digestive enzymes
Answer 41: (b)
42. Identify the two components of Phloem tissue that help in transportation of food in plants
(a) Phloem parenchyma & sieve tubes
(b) Sieve tubes & companion cell.
(c) Phloem parenchyma & companion cell.
(d) Phloem fibres and sieve tubes
Answer 42: (b)
43. A converging lens forms a three times magnified image of an object, which can be take on a screen. If the focal length of the lens is 30 cm, then the distance of the object from the lens is
(a) - 55 cm
(b) - 50 cm
(c) - 45 cm
(d) - 40 cm
Answer 43: (d)
44.
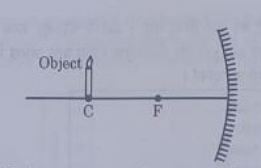
which of the following statement is not true in reference to the diagram shown above?
(a) Image formed is real.
(b) Image formed is enlarged.
(c) Image is formed at a distance equal to double the focal length.
(d) Image formed is inverted.
Answer 44: (b)
45.
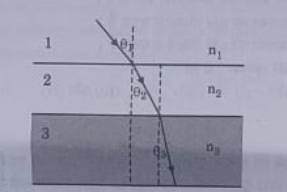
In the diagram shown above n1, n2 and n3 are refractive indices of the media 1, 2 and 3 respectively. Which one of the following is true in this
(a) n1 - n2
(b) n1 > n2
(c) n2 > n3
(d) n3 > n1
Answer 45: (d)
46. The refractive index of medium A is 1.5 and that of medium B is 1.33. If the speed of light in air is 3 x 108 m/s, what is the speed of light in medium A and B respectively?
(a) 2 x 108 m/s and 1.33 x 108 m/s
(b) 1.33 x 108 m/s and 2x 108 m/s
(c) 2.25 x 108 m/s and 2 x 108 m/s
(d) 2 x 108 m/s and 2.25 x 108 m/s
Answer 46: (d)
47. An object of height 4 cm is kept at a distance of 80 cm from the pole af a diverging mirror. If the focal length of the mirror is 10 cm. the height of the image formed is
(a) + 3.0 cm
(b) + 2.5 cm
(c) + 1.0 cm
(d) + 0.75 cm
Answer 47: (c)
48. 50.0 mL of tap water was taken in a beaker. Hydrochloric acid was added drop by drop to water. The temperature and pH of the solution wa noted. The following graph was obtained. Choose the correct statement related to this activity.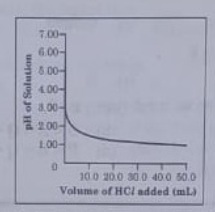
(i) The process of dissolving an acid an water is highly endothermic.
(ii The pH of the solution increases rapidly on addition of acid.
(iii) The pH of the solution decreases rapidly on addition of acid.
(iv) The pH of tap water was around 7.0.
Answer 48: (c)
Section-C consists of three cases followed by questions.
There are a total of 12 questions (Q. No. 49 to 60) in this section. Attempt any 10 questions from this section. The first attempted 10 question would be evaluated.
Case-I:
A student, took four metal P, Q, R and S and earned out different experiments to study the properties of metals. Some of the observations were:
• All metals could not be cut with knife except metal R.
• Metal P combined with oxygen to form an oxide M2O3 which reacted with both acids and bases.
• Reaction with water.
P — Did not react either with cold or hot water but reacted with steam
Q — Reacted with hot water and the metal started floating
R — Reacted violently with cold water.
S — Did not react with water at all.
Based on the above observations answer the following:
49. Out of the given metals, the one which needs to be stored used Kerosene is
(a) P
(b) R
(c) S
(d) Q
Answer: (b)
50. Out of the given metals, the metal Q is
(a) Iron
(b) Zinc
(c) Potassium
(d) Magnesium
Answer: (d)
51. Metal which forms amphoteric oxides is
(a) P
(b) Q
(c) R
(d) S
Answer: (a)
52. The increasing order of the reactivity of the four metals is:
(a) P<Q<R<S
(b) S<R<Q<P
(c) S<P<Q<R
(d) P<R<Q<S
Case-II:
The figure shown below represents a common type of dialysis called as Haemodialysis. It removes waste products from the blood. Such RB excess salts, and urea which are insufficiently removed by the kidney in patients with kidney failure. During the procedure, the patient’s blood is cleaned by filtration through a series of semi-permeable membranes before being returned to the blood of the patient. On the basis of this, answer the following questions: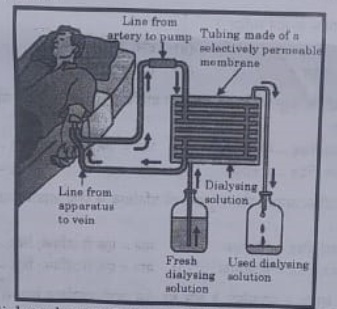
Answer: (c)
53. The haemodialyzer has semi-permeable lining of tubes which help to:
(a) To maintain osmotic pressure of blood.
(b) To filter nitrogenous wastes from the dialyzing solution.
(c) In passing the waste product, in the dialyzing solution.
(d) To pump purified blood back into the body of the patient.
Answer: (c)
54. which one of the following is not a function of Artificial Kidney?
(a) To remove nitrogenous wastes from the blood.
(b) To remove excess fluids from the blood.
(c) To reabsorb essential nutrients from the blood.
(d) To filter and purify the blood.
Answer: (c)
55. The 'used dialysing' solution is rich in:
(a) Urea and excess salts
(b) Blood cells
(c) Lymph
(d) Proteins
Answer (a)
56. Which part of the nephron in human kidney, serves the function of reabsorption of certain substances?
(a) Glomerulus
(b) Bowmans Capsule
(c) Tubules
(d) collecting duct
Answer: (c)
Case-III:
A compound microscope is an instrument which consists of two lenses L1 and L2. The lens L1 called objective, forms a real, inverted and magnified image of the given object. This serves as the object for the second lens L2; the eye piece The eye piece function like a simple microscope or magnifier. It produces the final image, which is inverted with respect to the original object, enlarged and virtual.
57. What types of lenses must be L1 and L2?
(a) Both concave
(b) Both convex
(c) L1 — concave and L2 — convex
(d) L1 — convex and L2 — concave
Answer: (b)
58. What is the value and sign of magnification (according to the new Cartesian sign convention) of the image formed by L1?
(a) Value = Less than 1 and Sign = Positive
(b) value = More than 1 and Sign = Positive
(e) value = Less than 1 and Sign = Negative
(d) value = More than 1 and Sign = Negative
Answer: (d)
59. What is the value and sign of (according to new Cartesian sign convention) magnification of the image formed by L2?
(a) value = Less than 1 and Sign = Positive
(b) value = More than 1 and Sign = Positive
(c) Value = Less than 1 and Sign = Negative
(d) Value = More than 1 and Sign = Negative
Answer: (b)
60. If power of the eyepiece (L2) is 5 diopters and it forms an image at a distance of 80 cm from its optical centre, at what distance should the object be?
(a) 12 cm
(b) 16 cm
(c) 18 cm
(d) 20 cm
Answer: (b)
Disclaimer: CBSE Class 10 Science answer key available here is for reference purposes and is not final. It is expected that the Official CBSE Class 10 Science answer key will be published by the board after the declaration of the result. Precautions have been taken while compiling the answers of this paper. However, there might be some questions & answers with some issues (for example: where more than one option is correct, no option is correct, etc.). Students are advised to discuss with their CBSE school teachers regarding the same before reaching a final conclusion.
Comments
All Comments (0)
Join the conversation